 
In both cases, one of the elements is a diatomic molecule because that is the standard state for that particular element. The formation reaction for carbon dioxide (CO 2) is For example, the formation reaction for methane (CH 4) is The product is one mole of substance, which may require that coefficients on the reactant side be fractional (a change from our normal insistence that all coefficients be whole numbers). By standard states we mean as a diatomic molecule if that is how the element exists and the proper phase at normal temperatures (typically room temperature). We need measure only the enthalpy changes of certain benchmark reactions and then use these reactions to algebraically construct any possible reaction and combine the enthalpies of the benchmark reactions accordingly.īut what are the benchmark reactions? We need to have some agreed-on sets of reactions that provide the central data for any thermochemical equation.įormation reactions are chemical reactions that form one mole of a substance from its constituent elements in their standard states. This is a very useful tool because now we don’t have to measure the enthalpy changes of every possible reaction. Hess’s law allows us to construct new chemical reactions and predict what their enthalpies of reaction will be.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

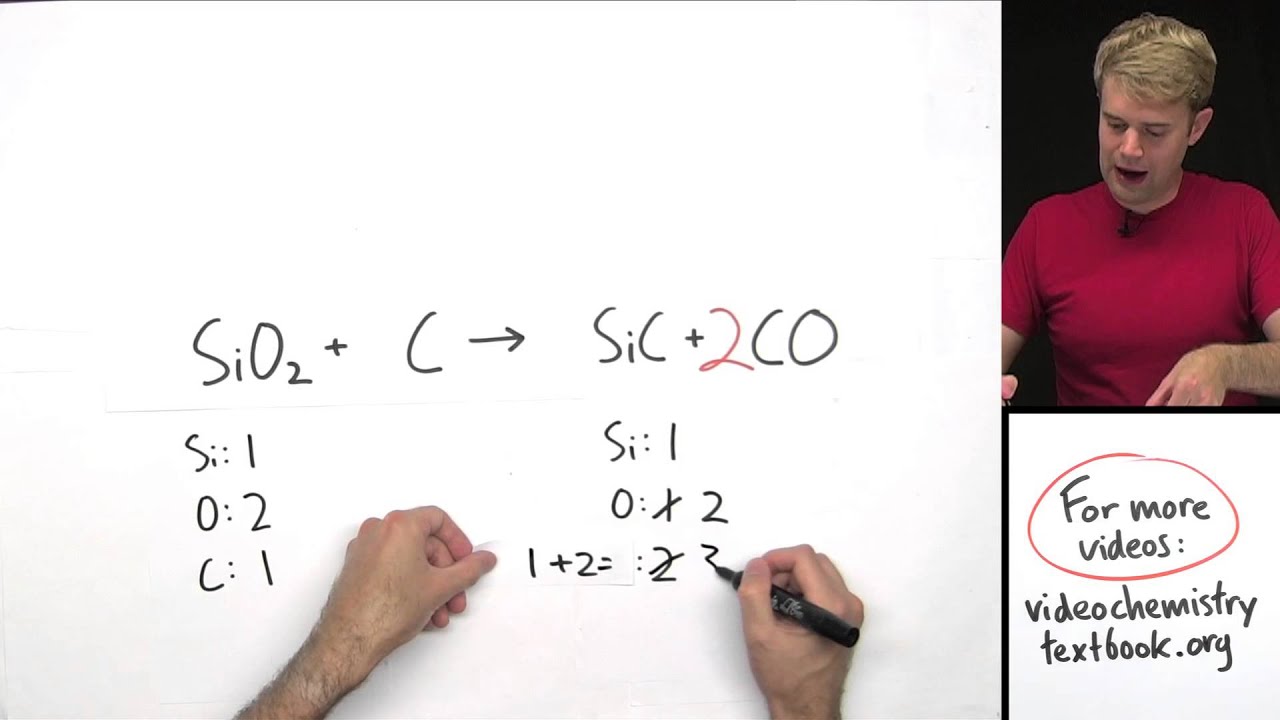
 RSS Feed
RSS Feed
